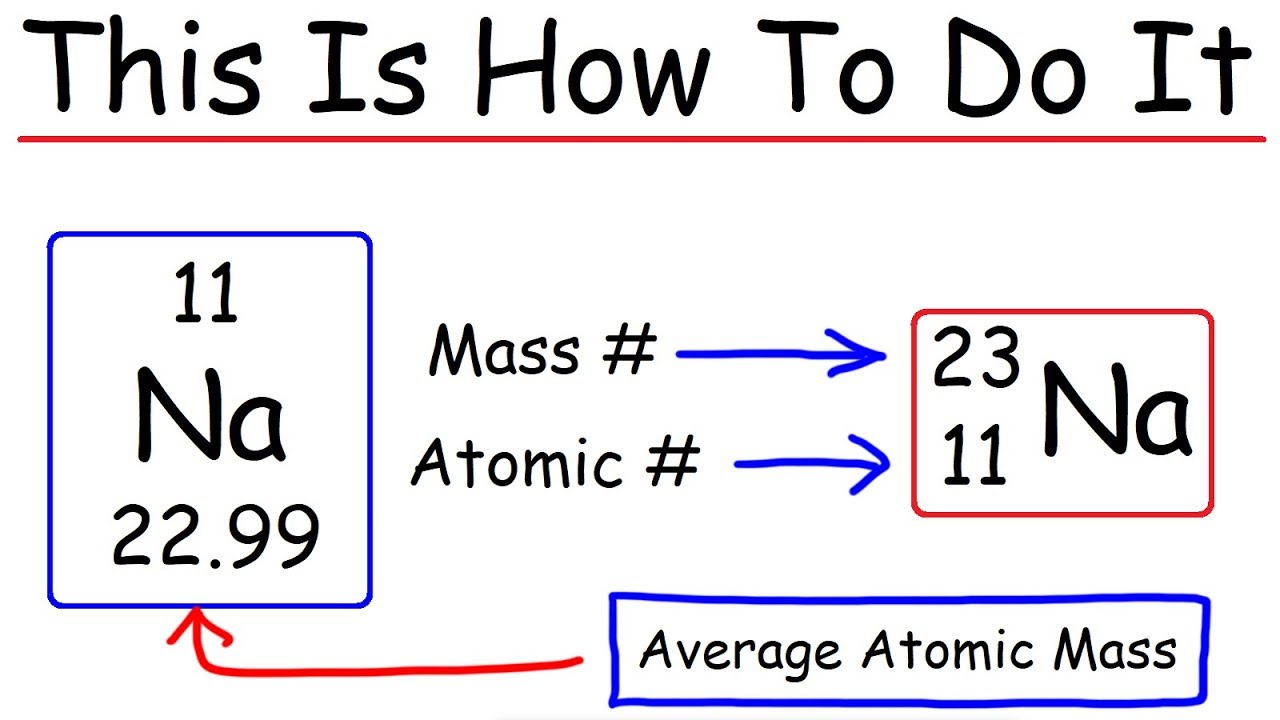
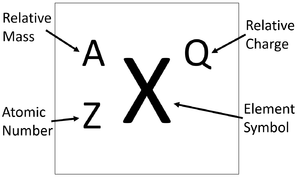
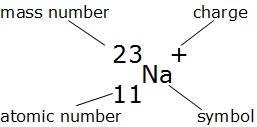
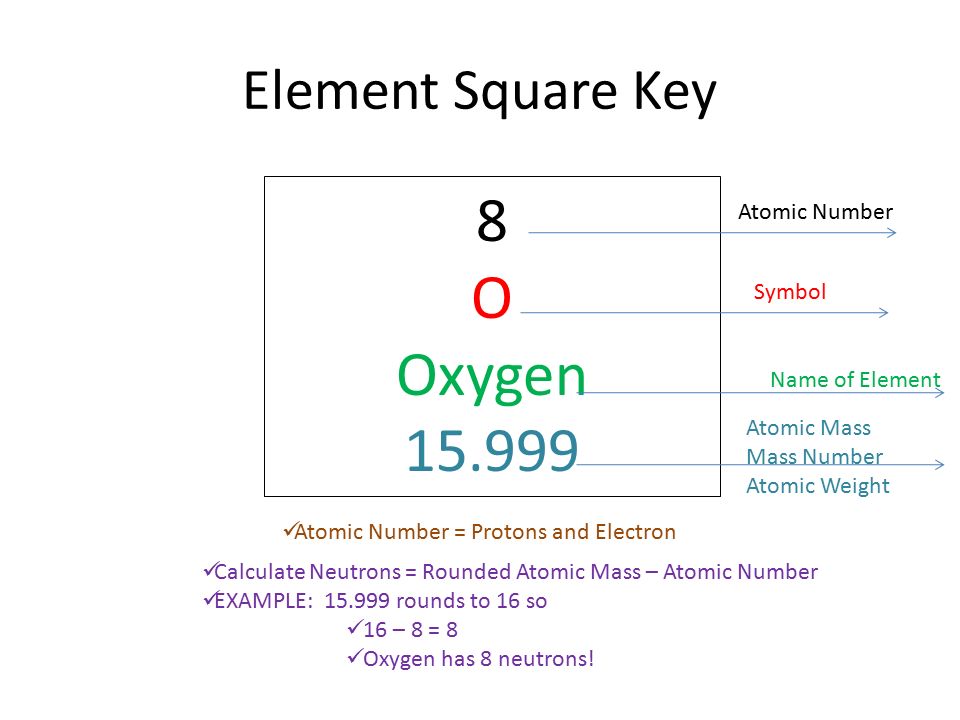
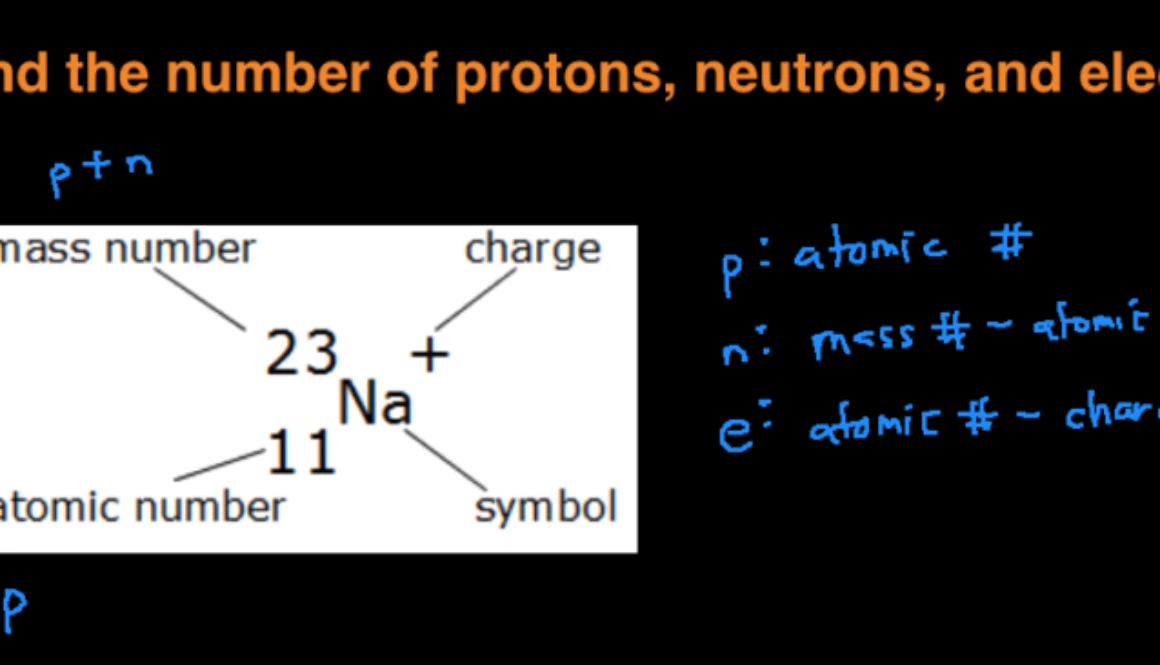
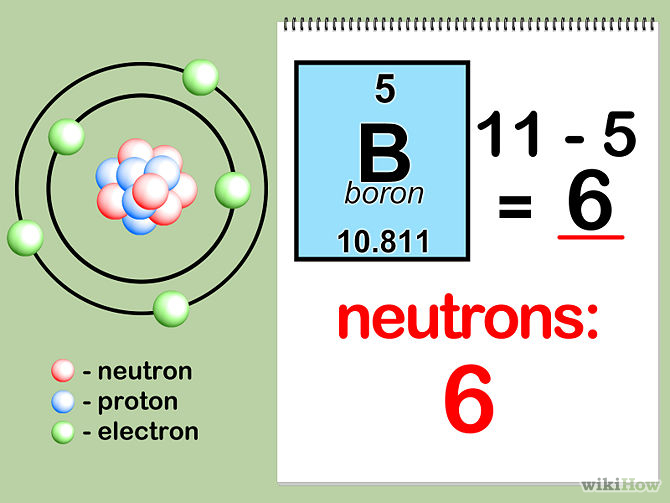
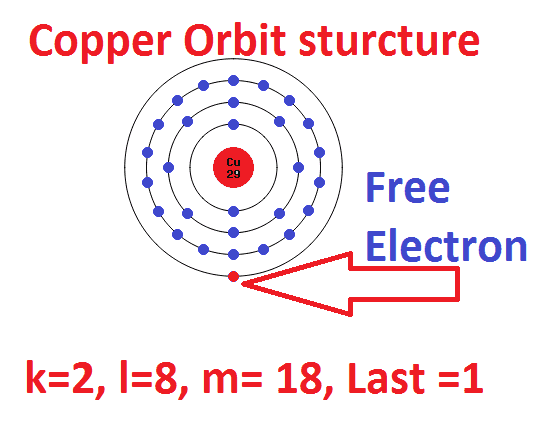
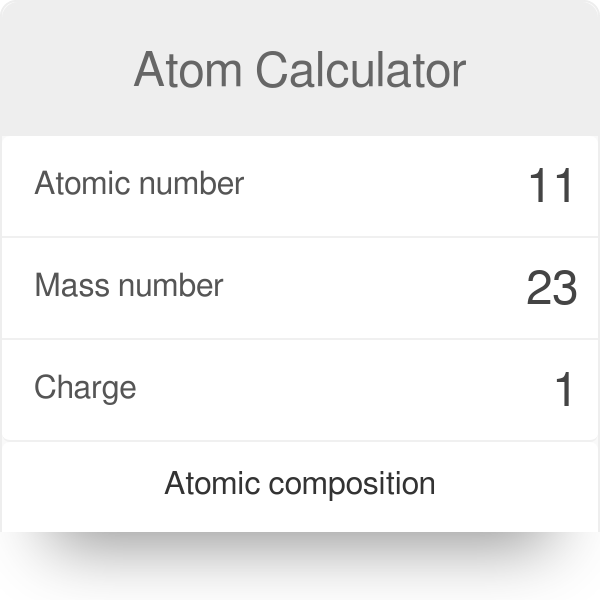
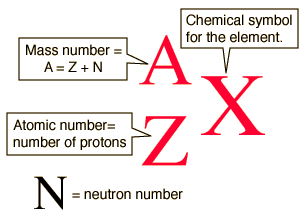Calculate the number of protons , electrons and neutrons in the following. (a) Chloride ion (Cl^(-)) with Z = 17,A =35 (b) Aluminium ions(Al^(3+)) with Z = 13 , A=27

Protons Neutrons Electrons Isotopes - Average Mass Number & Atomic Structure - Atoms vs Ions - YouTube