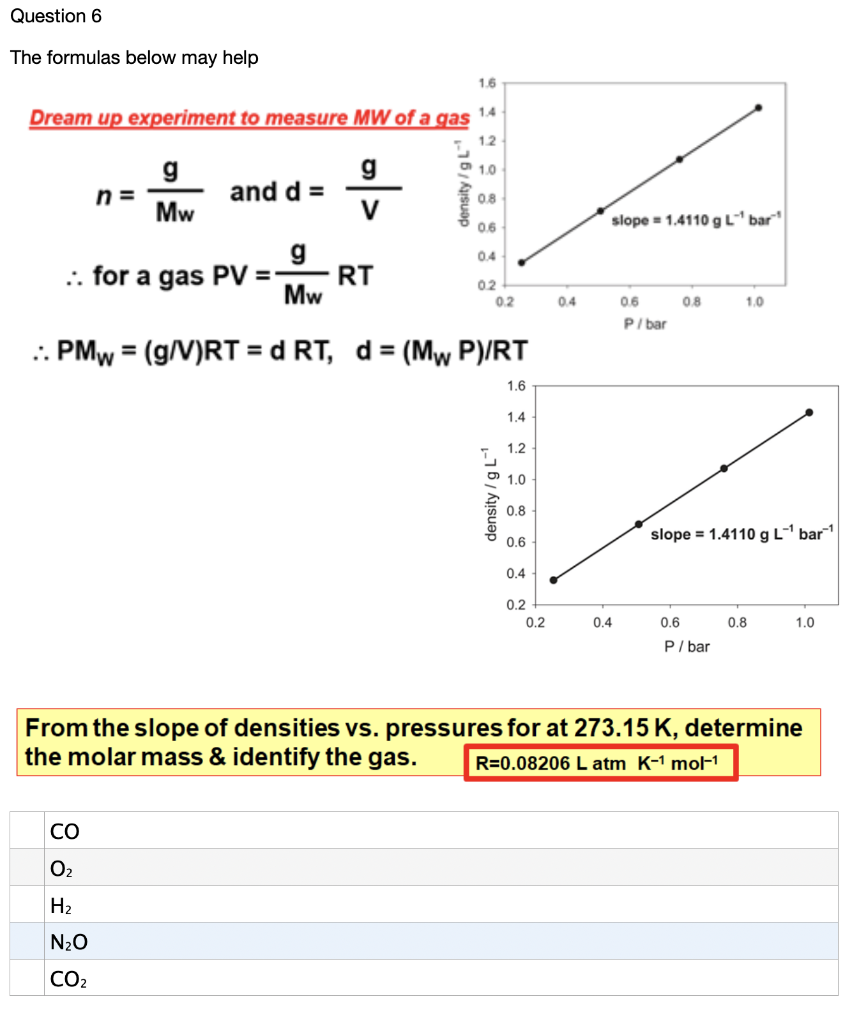
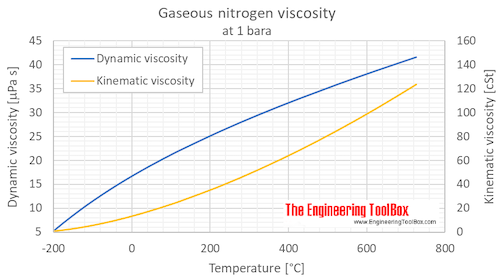
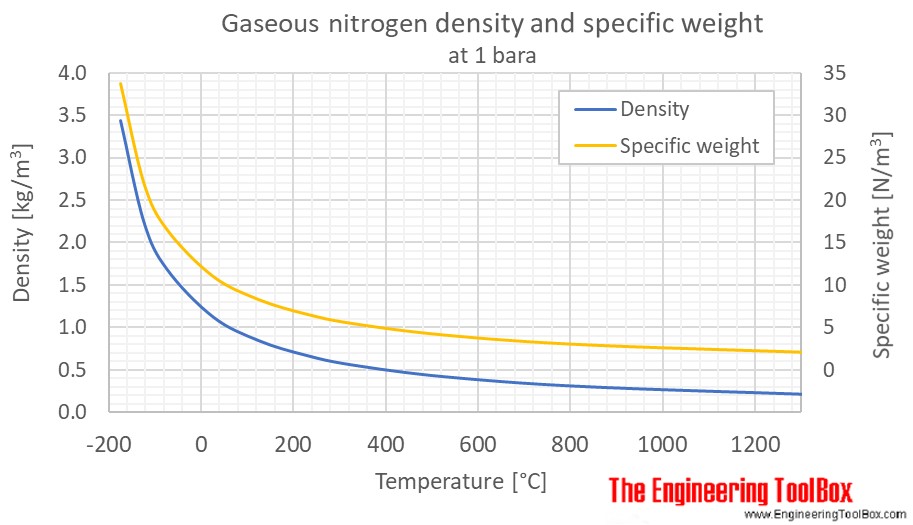
Calculate the volume at N.T.P occupied by (i) 14 g of nitrogen (ii) 1.5 gram moles of carbon dioxide (iii) 10^(21) molecules of oxygen.
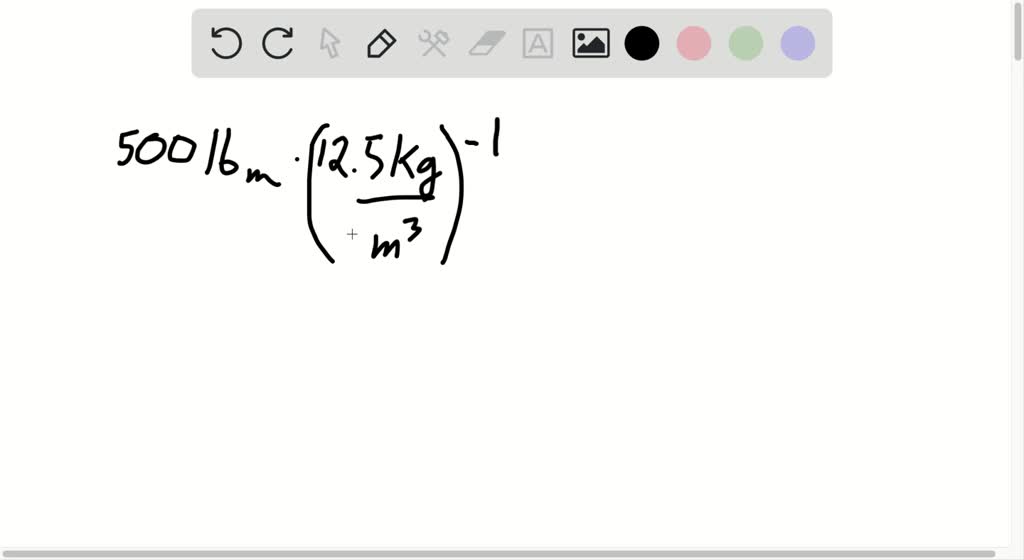
SOLVED:Five hundred 1 bm of nitrogen is to be charged into a small metal cylinder at 25^∘ C, at a pressure such that the gas density is 12.5 kg / m^3. Without